The article was written by Dr. Aydar Akchurin
High Hardness Boron Rich Boron Carbide is Synthesized using CVD
Hardness is a material’s resistance to plastic deformation and this property is widely used to rank materials for various engineering applications. Hard materials can be used for cutting, drilling. Hardness frequently correlates with wear resistance and the hardest material – diamond – is widely used in the applications requiring high wear resistance. It may be surprising, but three-quarters of the world’s supply of natural diamonds is used for industrial purposes (and only one third for jewelry).
Unfortunately, diamond has its limitations and the cost is not the only one. Diamond is prone to oxidation at high temperatures (over 600 degrees Celsius) and tends to chemically react with ferrous metals. This leaded many researchers to look for an (better) alternative material, that is equally hard, but also can sustain high pressure, temperature and corrosion. Most of the research in this area is concentrated over various materials containing C, N, B, O elements. Typically, these elements generate short covalent bonds with certain directionality, which makes them hard to deform. Hence materials made of these elements tend to have high hardnesses.
Since diamond is made of carbon and is known to be the hardest material, many of the newly thought and synthesized materials contain carbon. For example, carbon rich materials containing boron (diamond lattice can only take 7.7 at % of boron) are known to exhibit some attractive properties. In contrast, boron rich materials containing some carbon are not yet well explored.
A group of Professor Yogesh Vohra, University of Alabama at Birmingham, has developed a method to produce boron rich carbide ( ![]() ) films using microwave-plasma chemical vapor deposition (CVD). The use of CVD allowed the researchers to improve the control over impurities during production and increase the area of the produced films as compared to the existing methods.
) films using microwave-plasma chemical vapor deposition (CVD). The use of CVD allowed the researchers to improve the control over impurities during production and increase the area of the produced films as compared to the existing methods.
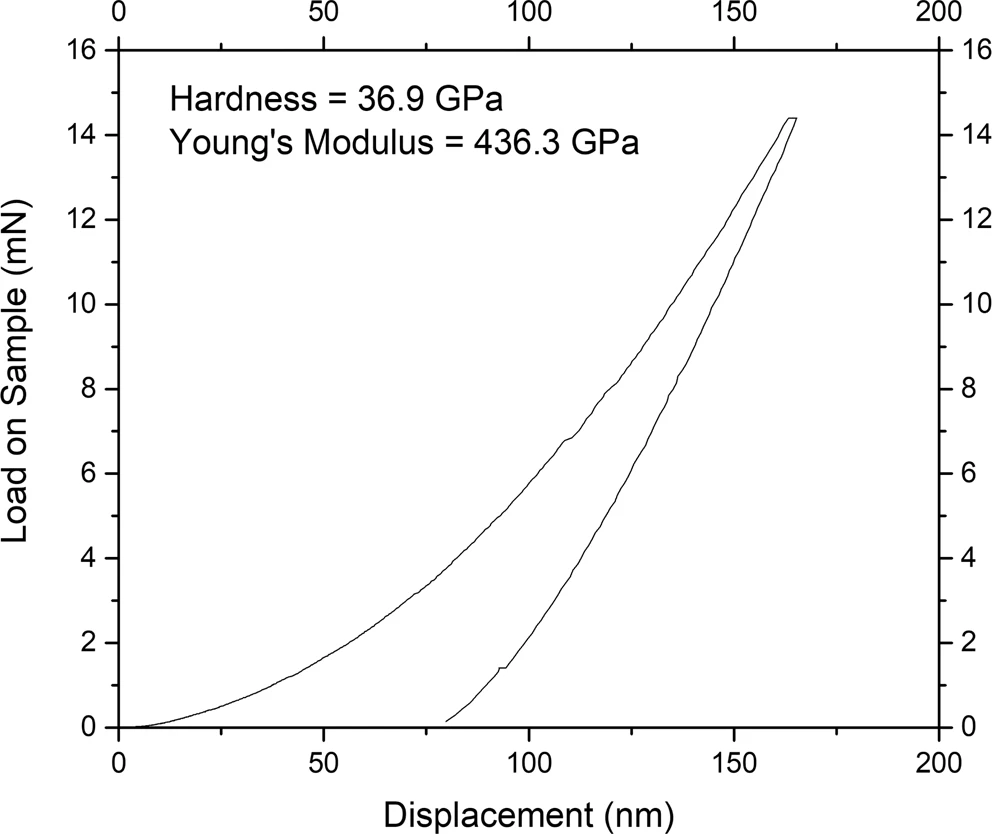
Boron rich carbide films were grown on a 550nm Silicon wafer substrate using 2.45 GHz microwave-plasma chemical vapor deposition system under 750 degrees Celsius temperature, at a pressure of 15 Torr (equivalent to the atmospheric pressure 15 miles above Earth) using 1 kW of microwave power for 4 hours. The deposited films were characterized using various techniques, including a nanoindenter. Nanoindentation measurements revealed the average hardness to be of around 14 GPa, however, reaching the values as high as 37 GPa. The large variations in the hardness values are attributed to the variation of the local geometry as can be seen in the SEM image of the film (see the featured image above). The measured hardness of ![]() is 37% of the hardness of diamond while Young’s modulus is about 41% of the value for the diamond.
is 37% of the hardness of diamond while Young’s modulus is about 41% of the value for the diamond.
Besides extensive experimental work, the researchers conducted first principles calculations (DFT) to get further details regarding the newly obtained material. From the simulations it was found that the carbon position in the lattice plays the most important role in changing the stability of the material. Positioning of carbon atoms in other locations was predicted to be unstable and resulting in generation of metallic materials. The proposed DFT model predicted the hardness as well as the electronic behavior of the boron rich carbide ![]() .
.
Further information: Paul A. Baker et al, First-Principles Predictions and Synthesis of B50C2 by Chemical Vapor Deposition, Scientific Reports (2020). DOI: 10.1038/s41598-020-61462-9


Be the first to comment