I am currently working as a Postgraduate Researcher at the University of Leeds, where I am actively involved in research activities. Prior to this, I successfully completed my master's degree through the renowned Erasmus Mundus joint program, specializing in Tribology and Bachelor's degree in Mechanical Engineering from VTU in Belgaum, India. Further I handle the social media pages for Tribonet and I have my youtube channel Tribo Geek.
Raman spectroscopy
Table of Contents
Introduction
The interaction of light on materials is very different it may be transmitted, reflected, or scattered; the wavelength of the light affects the interaction with materials in different colors. This study of light is called spectroscopy. Based on this an Indian physicist C.V Raman observed the scattering phenomenon where the light is scattered by the molecules and hence this phenomenon was named Raman scattering. The analysis/characterization technique that deals with Raman scattering is Raman spectroscopy. In Fig-1 Raman spectrometer from S & N lab is shown.

Fig-1 Raman spectrometer from S & N lab [1]
Definition
Raman spectroscopy is the analytical technique where scattered light is used to measure the vibrational energy modes of the sample. This technique provides both the information on chemical and structural characteristics of the material and also the identification of substances. The Raman spectroscopy extracts the information through the detection of Raman scattering from the sample. Fig-2 is the schematic representation of the Raman spectrometer.
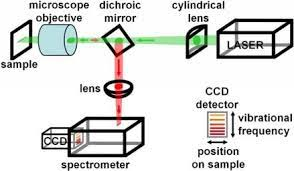
Fig-2 Schematic representation of Raman spectrometer [2]
Working principle
The working principle of Raman spectroscopy is based on the inelastic scattering of monochromatic light from a laser source which changes its frequency upon interaction with the material. Photons from the laser are absorbed by the samples and it is remitted with a frequency shift up or down in comparison to the original monochromatic frequency this is called the Raman effect. These shifts in the frequency provide information about the rotational, vibrational, and other low-frequency transitions in the molecules. This technique can be used in studying the materials like solid, liquid, and gaseous nature. In order to understand spectroscopy better, we should know the difference between Rayleigh scattering and Raman scattering.
- Rayleigh scattering: In this case, the energy of the molecules is unchanged after the interaction with the molecules. The energy and the wavelength of the scattered photons are equal to that of the incident photon. Hence the energy of the scattering particle is conserved this is called Rayleigh scattering.
- Raman scattering: In this case, the light is scattered by the molecule, and the oscillating electromagnetic field of a photon induces a polarisation of the molecular electron cloud causing the molecules to be in a higher energy state with the energy of a photon is transferred to the molecule. This can be considered as the formation of a very short-lived complex between the photons and molecules which is commonly called the virtual state of molecules. The virtual state is not stable, and the photon is remitted almost immediately as scattered light. The schematic representation of the Raman and Rayleigh scattering is shown in Fig-3.
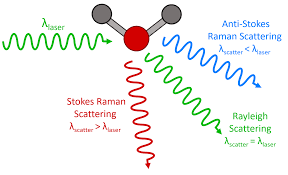
Fig-3 Raman scattering and Rayleigh scattering [3]
Components of Raman spectrometer
- Laser source: The laser source is used for the excitation of the sample and resulting scattered light.
- Injection/rejection filter: The filter delivers the laser to the sample and allows the scattered Raman light to pass through to the spectrograph.
- Spectrograph: The spectrograph is used to divide the light into separated wavelengths and measure the light intensity at each wavelength.
- Microscope: The microscope is used to focus the laser light onto a point on the sample surface and collects the Raman light.
- Computer: It provides instrumental control and data handling and manipulation.
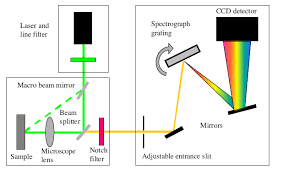
Fig-4 Schematic representation of Raman spectrometer with its components [4]
Information from Raman spectroscopy
The information that is obtained from the Raman spectroscopy is useful in analyzing various aspects of the material compositions. The Raman shifts and relative intensities of all Raman bands of the material allow identifying the material. The individual band changes and shifts which are seen as narrow, or broad can be varied with the intensity of the light. These changes can reveal information about the stresses in the sample and variation in crystallinity. The amount of material and its composition can also be identified, the variations in spectra with the position of the samples also reveal the changes in the material’s homogeneity.
Advantages and disadvantages
The advantages of Raman spectroscopy include its strength in specifying the chemicals in the materials which is a chemical fingerprint technique. There is no need for sample preparation and it is a non-destructive technique. The Raman spectra are acquired within a few seconds decreasing the processing time.
The disadvantages of Raman spectroscopy include that it can not be used in analyzing metals and alloys, and in most cases, it is not quantitative regarding the composition. The Raman effect is weak and the detection needs a very sensitive and highly optimized instrument. The fluorescence of impurities or of the sample itself can hide the Raman spectrum.
Reference
[1] http://www.snlabs.com/raman-spectroscopy.html
[2] Downes, A. and Elfick, A., 2010. Raman spectroscopy and related techniques in biomedicine. Sensors, 10(3), pp.1871-1889.
[3] https://www.edinst.com/blog/what-is-raman-spectroscopy/
[4] https://www.sas.upenn.edu/~crulli/TheRamanSpectrophotometer.html
Leave a Reply
You must be logged in to post a comment.

Dear Sirs
This is MPS kOREA.
I’m looking for Raman spectrometer for buying.
Can I get your specifications?
Regards
YH YI