Administration of the project
Sandia Researchers Identified Nanoscale Mechanisms of Steel Corrosion
Corrosion is a naturally occurring process, which converts refined metals into a more chemically stable form. This could be an oxide, hydroxide, or sulfide. Corrosion is the gradual destruction of metals by chemical or electrochemical reactions with the environment. Steel pipes are all subject to the inevitability of corrosion, resulting in oil companies spending thousands upon thousands of dollars to determine where pipes will need to be replaced before they experience complete corrosion and spill their contents into the surround environment.
![C:\Users\Melanie\AppData\Local\Microsoft\Windows\INetCache\IE\ATTTKPN1\Fe_corrosion[1].png](https://www.tribonet.org/wp-content/uploads/2019/07/c-users-melanie-appdata-local-microsoft-windows-i.png)
Researchers from Sandia National Laboratories have discovered a type of nanoscale corrosion, which is responsible for shortening the life span of steel pipes in an unpredictable manner.
Corrosion damage costs companies and governments trillions of dollars every year, increasing direct costs due to infrastructure and production and manufacturing and indirect costs due to cost of labour to investigate corrosion issues and loss of revenue. Oil spills are extremely detrimental to the environment. Consequently, the ability to reduce, or even better yet, stop, the corrosion process is a worldwide concern.
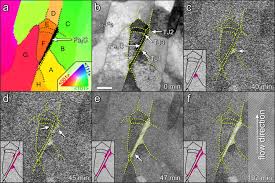
Research was conducted regarding oil pipelines and corrosion using transmission electron microscopes. As these microscopes are obviously too delicate to be used in the field, very thin samples of pipe were exposed at the research facility to various chemicals. These chemicals can all be found flowing through various pipelines. This lead to the creation of maps regarding types of steel grain and their orientation. Comparing maps from before the corrosion experiments to those after the experiments were conducted allowed the researchers to identify the weakest areas in the steel, which would therefore have the greatest propensity to be subject to corrosion.
Specifically, the transmission electron microscopes provided the means to identify the area of concern, which was discovered to be the spot where a compound composed of carbon and iron meets two grains of ferrite, which is a type of iron. This often occurs during the manufacturing of steel pipe. Unfortunately, these spots create a weaker area where corrosion easily occurs. Testing proved that the corrosion process halted when the area was completely eaten away. However, the process left behind a hole through which the corrosive solution is able to attack the interior of the steel.
This corrosion is an example of localized corrosion, which is the removal of metal by corrosion in small areas. Uniform corrosion proceeds at the same rate over the entire exposed metal surface. Uniform corrosion is predictable, while localized corrosion is not. Localized corrosion also opens up areas for uniform corrosion to move in and speed up the corrosion process. As a result, researchers conclude that understanding this process of localized corrosion is vital in the battle to prevent future catastrophes.
The research has several positive outcomes. The first is that the components involved in the initiation and progression of localized steel corrosion have been identified. This has lead to the creation of models designed to prevent future catastrophes such as pipeline beaks due to corrosion. The second outcome is that the researchers believe they have found a possible way to create new pipe, resistant to the corrosion process, by changing the microstructure of the steel during the forging process. Further testing needs to be conducted to confirm this idea, which will hopefully prove to be true. Pipelines more resistant to corrosion could ultimately save thousands of hectares of Earth’s environment.
Further information: Steven C. Hayden et al. Localized corrosion of low-carbon steel at the nanoscale, npj Materials Degradation (2019). DOI: 10.1038/s41529-019-0078-1
References:
- Image source.

Be the first to comment