Administration of the project
Speeding up electric vehicle battery charging
By Dr. Neil Canter, Contributing Editor | TLT Tech Beat November 2023
The rate of battery charging has been found not to be dependent on particle size.
• Fast charging of battery electric vehicles is a concern to consumers and was assumed to involve lithium particles staying intact when entering the electrode surface and diffusing into the bulk at a rate based on their size.
• A new approach, developed to evaluate how fast individual battery particles charge and discharge, demonstrated that smaller particles charge at the same rate as larger ones.
• The reason that particle size is not a factor may be due to the ability of liquid electrolyte to penetrate into cracks and break in the lithium particles facilitating the process.
The leading commercial technology used in battery electric vehicles is lithium-ion batteries. In a previous TLT article,1 researchers developed a lithium-air battery that potentially exhibits an energy density much beyond what is possible for lithium-ion battery technology. Lithium ions originating at the anode react with oxygen from the air at the cathode to produce energy. The lithium-air battery generates energy not through an intercalation process but by chemical transformation. Using a pathway that produces lithium oxide, a four-electron reaction can be achieved leading to the higher energy density.
One of the other concerns about lithium-ion batteries is the rate of charging. Consumers would like to charge a battery at the same speed as pumping gasoline or diesel into an internal combustion-powered automobile. Yiyang Li, assistant professor of materials science and engineering at the University of Michigan in Ann Arbor, Mich., says, “The most widely used lithium-ion battery contains a NMC-type cathode prepared from oxides of lithium, nickel, manganese and cobalt. Primary and secondary particles are present in the polycrystalline cathode. The former have diameters of approximately 10 microns and agglomerate into secondary particles that range in size from 1,100 to 500 nanometers.”
Li indicates that the charge/discharge process is the reversible reaction that moves lithium between a cathode and a graphite anode. When the first lithium-ion batteries were introduced for portable electronic applications, they used single-crystal lithium cobalt oxide particles. These monolithic single crystals do not break apart easily, so it is established that lithium enters the particle at the surface and diffuses into the bulk.
Li says, “For electric vehicles, lithium cobalt oxide is too expensive, so most of the cobalt was replaced with nickel and manganese, which are much more abundant, and less expensive minerals. In addition, the single-crystal particles were replaced with polycrystalline particles, which look more like meatball particles that could potentially break apart. Despite this change in the material, it was still believed that the particle stays intact, and lithium can only enter at the surface and diffuse into the bulk.”
A schematic showing this standard model can be found in Figure 2a. Since most battery experiments measure the average behavior of billions of particles, it has been difficult to validate or disprove this assumption.
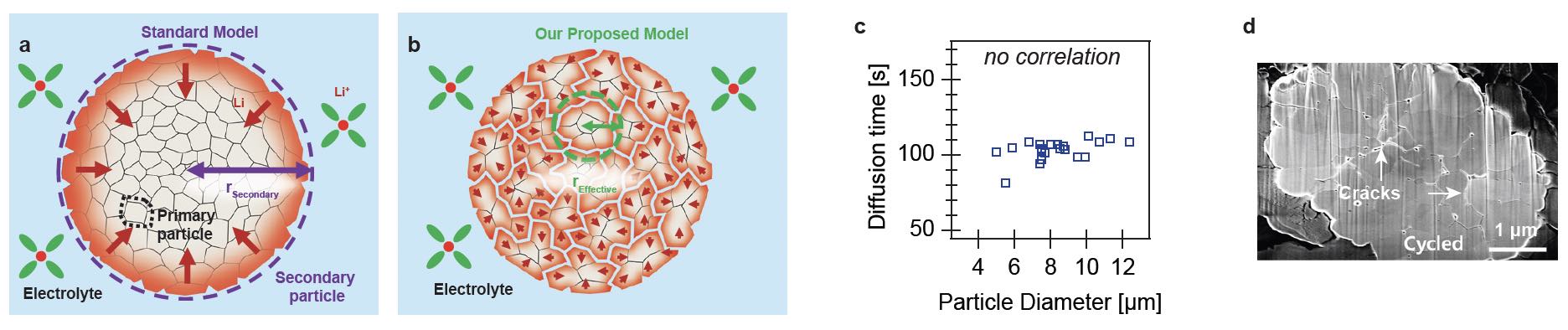
Figure 2. The currently accepted model for battery charging is shown in Figure 2a where particles entering the bulk diffuse at a rate based on their size. After using a new approach to better understand battery charging, the researchers propose a different model in which electrolyte penetration into battery particles impacts the rate of battery charging as illustrated in Figure 2b. The graph in Figure 2c shows no correlation between particle size and diffusion rate, which counters the currently accepted model. Evidence of cracks and breaks in a particle is shown in the scanning electron microscope image in Figure 2d. Figure courtesy of the University of Michigan.
Li and his colleagues have now developed a new approach, borrowed from neuroscience, to evaluate this theory experimentally by charging and discharging individual battery particles.
Intergranular cracking
The researchers charged and discharged individual particles by preparing a multi-electrode array that contains lithographically patterned gold microelectrodes, 20 microns in size. Li says, “We were successfully able to charge and discharge individual battery particles using this design, which is very similar to what is done to measure extracellular field potentials from mammalian neurons.”
The researchers then evaluated how fast the particles charged. Li says, “To our surprise, smaller particles charged no faster than larger ones. This result challenges one of the textbook descriptions of battery charging. The evaluation done was not conducted on an obscure battery material, but in fact describes the most common cathode material used in electric vehicles today.”
The researchers duplicated this result multiple times and concluded that smaller particles charge at the same rate as larger particles. Figure 2c shows plots of diffusion time versus secondary particle diameter. It is expected that larger particles would charge slower; instead, there is no relationship between particle size and charging rate.
To explain this finding, the researchers propose that the “meatball” particles are not monolithic, but in fact form cracks and breaks along the grain boundaries (see Figure 2d). As a result, the liquid electrolyte can penetrate the secondary particles (see Figure 2b). These pathways effectively act as highways for lithium transport into the particles, ensuring that no matter how large the particle, the lithium can quickly enter and leave, thereby enabling faster charging.
Li will be working with single-crystal cathode particles in future work. He says, “Single crystal particles exhibit longer cycling lives because they are believed to not crack; however, without these cracks, they also lack the highways that enable faster charging.”
Li wonders if this is the reason that single-crystal NMC particles will be very challenging. He says, “They may last for one million miles, but they may not be able to be supercharged.”
Additional information on this work can be found in a recent paper2 or by contacting Li at [email protected].
REFERENCES
1. Canter, N. (2023), “Lithium-air battery,” TLT, 79 (6), pp. 32-33. Available here.
2. Min, J., Gubow, L., Hargrave, R., Siegel, J. and Li, Y. (2023), “Direct measurements of size-independent lithium diffusion and reaction times in individual polycrystalline battery particles,” Energy & Environmental Science, 16 (9), pp. 3847-3859.
Neil Canter heads his own consulting company, Chemical Solutions, in Willow Grove, Pa. Ideas for Tech Beat can be submitted to him at [email protected].


Be the first to comment