Tribochemistry
Table of Contents
Boundary Lubrication
Boundary lubrication is the lubrication regime where the interface behaviour is dominated by chemical reactions that happen at the surfaces, tribofilm formation occurs, and the load is carried by the asperities. In the boundary lubrication regime the asperity-to-asperity contacts may lead to elastic or plastic deformation or even fracture and can generate frictional heat which will be accompanied by chemical reactions to produce organic and inorganic surface films (Figure 1). A wide range of studies regarding many aspects of tribofilm formation and removal and their roles in reducing friction and wear have been conducted.
The effectiveness of boundary lubrication has been considered for a long time as a necessity for modern designs of machines with reliable operations. Because of the need for more energy efficiency, availability of new materials and machine part downsizing, the need for understanding true interactions in this regime is of great importance. The boundary lubrication regime has been the subject of many studies for more than 70 years and the majority of these studies are experimental investigations into the nature of what happens in this regime. Many of the studies cover the boundary film chemical, physical and mechanical properties and their effects on wear and friction reduction. The subject of many works has been to investigate different kinds of additives in oils and their effects on various aspects of tribological performance. As the boundary lubrication regime is mainly related to interactions of two surfaces and the additive containing oils between them, the analytical studies of surfaces including topography measurements, chemical analyses, mechanical and physical studies are considerable. All these experiments give good insight into different chemical and physical characteristics covering various aspects of boundary lubrication systems.
It is clear from the wealth of experimental literature in this area that the nature of the phenomenon happening in this regime is very complicated. Studying the entire problem needs a multiscale understanding ranging from component scale down to the micro-scale and also molecular interactions of films and lubricant additives. Experimentation across such scales is challenging and hence it is important to complement such studies with the ability to predict the friction and wear of a working system without running experiments. It is also important to analyse the system and optimise its performance in order to design cost effective experiments. Many modelling attempts have been made in the past years but a comprehensive multiscale model of boundary lubrication considering tribochemistry phenomenon in order to predict friction and wear of the system is still lacking.
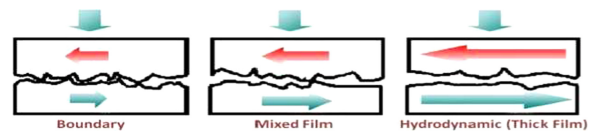
Figure 1 Different lubrication regimes
ZDDP as an anti-wear additive
Additives are vital elements for lubricants used in wide ranges of engineering applications including vehicles, turbines and production facilities. As the names imply, anti-wear (AW) additives and friction modifiers can improve the performance of many tribological systems by decreasing the wear and friction. ZDDP is one of most common AW additives applied in the various industries due to its outstanding ability to generate protective films named tribofilm on contacting surfaces. Apart from providing anti-wear properties, ZDDP can also work as a multifunctional additive showing antioxidant and anticorrosion performance. Although usage of ZDDP brings such benefits to the system, it has several drawbacks as well. The generated tribofilms were found to aggravate the micropitting in rolling contacts due to the generated high friction and low wear rate. The severity degree of micropitting increases with the thickness of tribofilm, which leads to the reduction of bearing service life. In addition, due to the presence of sulphur and phosphorus, application of ZDDP to the systems equipped with catalytic converters such as the exhaust system of vehicles can poison the catalyst producing harmful emissions although its performance highly relies on the two chemical elements discussed in the following section. As the strict rules on the limited usage of the ZDDP in oils are proposed, it is necessary to exploit any environmental-friendly alternative with similar performances. To completely replace the ZDDP, knowledge about the ZDDP derived tribofilm in terms of its wear mechanism, properties, the kinetics, its morphology and the relevant rheological study is necessary and is presented here.
Introduction to the ZDDP Derived Tribofilm
Currently tribofilm is widely recognized as the thin reacted film on substrate as a product of the tribochemical reactions at the interfaces driven by the friction heating and rubbing of contacting surface. It can be stress-induced at a temperature that is much lower than that required for thermal film due to the effect of rubbing on the reaction kinetics. The tribofilm was suggested to have an amorphous structure from a cross-section view by using the transmission electron microscopy (TEM). The multi-layered structure of tribofilm is verified by different researchers and shown in Figure 2, which has the rough, patchy and pad-like structure characteristics.

Figure 2 Schematic of the morphology of the ZDDP tribofilm. From Ref [1]
It consists mainly of a mixed Fe/Zn polyphosphate covered by the zinc polyphosphate with iron sulphide or zinc sulphide close to the steel substrate. The tribofilm can attain a relatively thick thickness ranging from 50 − 150 nm in the case of a steel substrate with different chemical natures from the substrate. By using X-ray adsorption near-edge structure (XANES), the chain of the glassy polyphosphate film was found to be longer on the top of tribofilm than that adjacent to the substrate interlinked with iron oxide. The reasons for the different lengths of chains were discussed by several researchers and different theories are summarised in the subsection ‘The Evolution of Polyphosphate’.
Wear Mechanism of the ZDDP Tribofilm
So far a certain consensus is reached on the origin of anti-wear properties of ZDDP additive, which is explained by the four mechanisms as follows: ZDDP forms the phosphate-rich layer showing the sacrificial nature at the contacting surface to reduce the direct contact of surfaces, thus preventing the adhesive wear of the rubbing surfaces and reducing the transient contact stresses the surfaces experience The phosphate layer can digest the hard and abrasive particles (mainly metal oxide) from the contacting surface, thus mitigating the three-body abrasive wear by producing more softer and benign iron sulphides at the region close to the substrate; ZDDP can not only reduce the surface oxidation by decomposing peroxy radicals but also reduce the corrosion by reducing peroxides in the base stock or a combination of any mechanisms above. Besides the four widely recognized anti-wear mechanisms, the specific effect of sulphur is considered in the wear reduction. Shimizu et al. [2] suggested that the extreme pressure response provided by the sulphur content of tribofilm plays an essential role in the protecting of substrate surface from initial wear while Martin [3] highlighted that the sulphur reacts with the nascent iron surfaces produced under severe wear condition to reduce adhesion and the attack of oxygen species. Besides, they suggested that for the mild-wear contact region, the anti-wear performance of tribofilm mainly drives from the rheological properties of zinc polyphosphate glasses. This specific anti-wear mechanism owing to the behaviour of the phosphate glassy structure similar to a viscous lubricant was also discussed by Morina et al. [4]. When it comes to more severe conditions, researchers found that the zinc phosphate glass can eliminate abrasive iron oxide particles based on the acid-base chemical reactions, which is similar with the second mechanism shown above. It is noted that the acid-base chemical reaction is an essential concept almost throughout the studies of Martin about tribofilm, which would be discussed in the following sections. Aside from the proposed anti-wear mechanisms above, the wear protection was suggested to result from the wear debris from tribofilm re-entering the contact zone. In addition, it was emphasized that the outstanding anti-wear properties of tribofilm result from not only the sufficiently high shear strength to maintain its integrity and adequate formation rate to generate continuous films but also a sufficiently low shear strength to ensure that the shear plane is located inside the tribofilm and the little part of applied load would be transmitted to the substrate material. In other word, it works like an impact guard for the automotive that absorbs the external impact energy and keep the distribution of impact energy outside the area where the passengers sit.
Kinetics of the ZDDP Tribofilm
Before the introduction of the tribofilm kinetics, the two new concepts ‘mechanochemistry’ and ‘in-situ’ are explained, which play the key role in the detailed analysis on the tribofilm formation and removal.
Mechanochemistry
An analytical method that considers the molecular behaviour and chemical reaction energy in terms of force and investigates the force-induced reactivity is called mechanochemistry. This view introduces the concept of stress-augmented thermal activation, which states that the activation energy barrier that regulates molecular or atomic processes can be reduced with the application of moderate stress as shown in Figure 3.

Figure 3 (a) Activation energy barrier without applied force on the molecule; (b) Reduction of energy barrier under the influence of applied force f. Figure from Ref [5]
To describe the augmentation process in detail, the applied mechanical force could promote the breaking of the molecular bonds. Similar with the role of the weakly-bonded atoms of surface mentioned in the subsection ‘Characteristics of Surface Texture’, these affected molecular bonds leads to higher reaction rate. Based on the theory, it has been long acknowledged that the rubbing of two contacting rough surfaces under boundary or mixed lubrication can accelerate the formation of tribofillm. The relevant study on this field is named tribochemistry. It is noted that the force stimulating the tribochemical activation is the shear stress instead of the normal pressure although the shear stress is approximately proportional to the pressure at a certain temperature in boundary lubrication. In addition, it had been discussed that other parameters including flash temperature, generation of catalytic surfaces, enhanced mixing of substrate due to shear and tribo-emission could also play the similar role. However, experiments by Zhang et al. [6] have shown that the tribochemical reaction for the formation of ZDDP tribofilm is promoted primarily by shear stress.
Ex-situ and In-situ Experiments
Compared with the traditional ex-situ experiments of the tribofilm formation, the in-situ ones are able to provide more precise information for the kinetic analysis as the information collection and dynamic growth of tribofilm are carried out concurrently. In comparison, ex-situ test suffers from the following limitations. Firstly, the detailed operating conditions of the surface analysis and dynamic test environment are different, which would possibly lead to the changing composition of the newly formed tribofilms due to its exposition to the new environment with different temperatures and humidity level. In addition, the surface could be contaminated by the adventitious entities, which would significantly affect the measurement results carried out later. Besides, the common practice before ex-situ study such as using solvents to rinse the surface would remove part of the tribofilm layers. Hence, some valuable information could be lost after this normal operation. Last but not least, the surface under the ex-situ study could not be probed until the tribological test is completed. Therefore, the real dynamic processes of the tribofilm formation and the existence of potential precursor or intermediate cannot be identified clearly. All above undermines the credibility of the experimental finding based on ex-situ tests.
The Evolution of Polyphosphate
To start the introduction of kinetics with the chain length evolution, it is a complicated process throughout the tribofilm formation. The debate on whether short-chain polyphosphates form initially and then are polymerised, or whether the long-chain polyphosphates form firstly and are truncated by cations and the rubbing process to form short-chain polyphosphates is presented. According to Martin et al. [3] , the oxygen present in the lubricant reacts with the nascent surface generating iron oxide particles under severe sliding motion, which are digested by the phosphate glass following the aforementioned acid-base reaction using the chemical hardness approach. The iron cations in the Fe 3+ oxidation state acts as the essential modifier for the network constituted by the phosphate chains in the proposed model. With the proceeding of the reaction, the chain length of the phosphate decreases where a depolymerisation process is involved. To explain it briefly, when the wear of metal surface occurs, iron oxides are formed. The generated hematite (Fe23) has the property of high hardness and melting point, which can be viewed as hard bases. Compared to it, phosphate glass of the tribofilm can be treated as hard acids. According to the hard soft acid and base (HSAB) principle, these two can have acid base reactions. The exchange of Zn2+ with Fe3+ needs more negative charges to balance the reaction, which can be obtained by shortening the chain length. This whole process is named depolymerisation. Given that there normally exists higher amount of wear particles at the side close to the steel surface, more depolymerisation happens bringing about shorter chain length. This specific ability of tribofilm to digest the existing iron oxide affects its oxygen content and is suggested to be one of the anti-wear properties of ZDDP with the developed molecular dynamics model [7]. Although this theory seems to makes sense, it lacks direct experimental evidence. In addition, it can hardly explain the tribofilm growth on nonferrous surfaces. On the contrary to the theory of Martin, Mosey et al. [8] argued that the cross-linking induced by high pressure based on the quantum chemical simulations is the reason for the chemically-connected networks of tribofilm with improved mechanical properties. The high pressure at the surface of the film is suggested to cause higher cross-linking of zinc phosphate molecules and result in longer-chain phosphate. Nevertheless, the pressures required were found to far exceed the yield pressures of most materials. Thus it is unlikely to happen in rubbing contact. High pressure infrared studies also showed no sign of structural change of ZDDP tribofilm even up to a pressure of 21 GPa.
Properties of the ZDDP Tribofilm
To illustrate the properties of tribofilm clearly, its mechanical properties and tribological properties, relation between its chemical properties and tenacity are discussed separately.
Mechanical Properties and Tribological properties
About the mechanical properties of ZDDP derived tribofilm, it is acknowledged that its hardness varies with its thickness by relevant nano-hardness experiments. As shown in Figure 4, the top layer (long chain polyphosphate) is found to be softer than the bottom layer (short-chain polyphosphate).

Figure 4 The varying mechanical properties of tribofilm (a) before and (b) after washing with solvent [9]
It could be said that the tribofilm accommodates the indentation pressure by increasing its hardness like a smart material. This hardness gradient is suggested to derive from the effect of iron in the phosphate, which is to harden the polyphosphate as explained in his theory of chain evolution. As to its tribological properties, it was found that there is a correlation between the formation of the thick tribofilm and the occurrence of high friction. As a result, the formed tribofilm was experimentally found to aggravate the micropitting in rolling contacts when the tribofilm provided excellent wear resistance. Ghanbarzadeh et al. [10] found that the growth of polyphosphate-rich film formed on the contacting asperities is responsible for an increases in the roughness of the surfaces in contact. They also indicated that the initial surface roughness of both contacting surfaces determines the later topography evolution due to the tribofilm growth. These properties of tribofilms were found to evolve during the period of tribofilm growth and removal with the initial stages characterized by a comparatively low hardness of a tribofilm which increases in hardness as the growth proceeds and eventually decreases in hardness since part of the tribofilm present on the surface are removed. Same with the ZDDP growth kinetics, the properties of tribofilm were found to vary with the operating conditions including the temperature and applied load.
Tenacity and the Relation with Chemical Properties
Tribofilm derived from ZDDP is proved to be relatively tenacious than that from other additives. Once formed, the tribofilm is stable and durable to withstand the further load. It is noted that the durability of tribofilm is also influenced by the temperature and applied load. Besides, the different chemical properties throughout the thickness of tribofilm determines the durability of tribofilm. Among the chemical substances, sulphide plays the key role to obtain a reliable tribofilm. Dorgham [11] stated that the sulphur-rich layer is the necessity for the formation of tenacious tribofilms on the rubbing surfaces based on his experimental finding that the base layer acts like a glue binding the substrate with the zinc phosphate layers that constitutes the main bulk of tribofilms, which corresponds to the finding of Mourhatch [12] saying that the ZDDP tribofilm attached to the substrate mainly through S − Fe bonds. According to Parsaeian et al [13], the chemical characteristics and durability of tribofilm develops in times. Longer chain polyphosphate were found to convert to the shorter one when severe rubbing occurs. This structural change leads to the increasing durability of tribofilm. In general, ZDDP tribofilm is less durable at the early stage of its formation period and the durability keeps evolving. Similar to the debate on the evolution of chain length, so far there is no consensus on the evolution of the sulphur species found in the ZDDP tribofilm.
Computational modelling in tribochemistry
Introduction to Numerical Methods
This subsection introduces briefly the main computational methods applied for the study of tribological problems in boundary lubrication including the molecular modelling (MD), finite element method (FEM), boundary element method (BEM) and discrete element method (DEM). A map of the representative tribological models built across the time and size scales is shown in Figure 5.

Figure 5 Schematics of developed models in different time and length scales for tribological problems. Figure from [14]
Figure 5 Represents models developed to solve tribological problems. MD is the simulation of physical movement of atoms and molecules. In MD models, the kinetics of atoms are determined by solving the Newtonian equations of motion for the interacted atoms and molecules. The forces between atoms and molecules are derived from the molecular mechanics force. In general, this method is effective to study the molecular interactions between the lubricant and substrates when the considered scale is extremely small. In consideration of the long reaction times as well as the large amounts of loading cycles for the tribofilm growth, this small-scale investigations can hardly capture the kinetics of tribofilm although it might be able to provide valuable information about the adhesive force between the tribofilm and surface asperity or the tribofilm with tribofilm on the molecular scale. DEM is based on the theory stating that the macroscopic behaviour of bulk is controlled by the microscopic behaviour of small particles. Similar to the MD, the Newtonian law is applied to determine the motion of particles and their interaction. This method assumes that at every time point the velocity of each particles is constant and the position of each particle could be updated. Given that the tribofilm is formed on the asperity due to the asperity-asperity or asperity-additive interactions, the information about microscopic scale behaviours of particles provided by DEM can be valuable. Besides, DEM can model the surface roughness with discrete particles and the wear of material by the displacement of particles form the built bond. However, DEM is also limited with its small time and length scale. Like MD, it can hardly capture the macroscopic behaviour of the system and there is problem of inevitable calibration of particle bonds. FEM is a well-known and a robust method to study the continuum mechanics. Although this method is widely applied in the study of solid mechanics simulations, it has several limitations in modelling the behaviour of the tribofilm. The analysed tribofilm is expected to modify the surface topography constantly along with the wear of materials and plastic deformation during its formation period. As a result, a reliable finite element model requires a constant variation of the mashing near the surfaces, which would increase the computational time considerably especially in the case when the model needs to mesh the entire contacting body instead of the specific contacting surface. Even though the computational efficiency is not neglected, the change of the surface roughness during the contact time can be too small for FEA to capture. Nevertheless, FEM is a robust approach for calculating the mechanics of contact thanks to its rigorous mathematical formulations. Compared to FEM, BEM is an efficient and easy method for surface characterization. Only the boundary of materials indicating the surface of contacting bodies needs to be meshed in this case indicating a two-dimensional spatial discretization, which saves computational time. Moreover, it handles the local contact properties properly, which is essential for the research on the local tribofilm growth in the boundary lubrication. Nevertheless, it is difficult to implement chemical analysis by BEM since a robust kinetics model as well mechanical properties of the thin films are needed.
References
- Spikes, H., The history and mechanisms of ZDDP. Tribology letters, 2004. 17(3): p. 469-489.
- Shimizu, Y. and H.A. Spikes, The tribofilm formation of ZDDP under reciprocating pure sliding conditions. Tribology Letters, 2016. 64(3): p. 46, https://doi.org/10.1007/s11249-016-0776-6.
- Martin, J., Lubricant additives and the chemistry of rubbing surfaces: metal dithiophosphates triboreaction films revisited. Japanese Journal of Tribology, 1997. 42(9): p. 1095.
- Morina, A. and A. Neville, Tribofilms: aspects of formation, stability and removal. Journal of Physics D: Applied Physics, 2007. 40(18): p. 5476.
- Spikes, H., Stress-augmented thermal activation: Tribology feels the force. Friction, 2018. 6(1): p. 1-31.
- Zhang, J. and H. Spikes, On the mechanism of ZDDP antiwear film formation. Tribology Letters, 2016. 63(2): p. 24, https://doi.org/10.1007/s11249-016-0706-7.
- Martin, J.M., et al., The origin of anti-wear chemistry of ZDDP. Faraday discussions, 2012. 156(1): p. 311-323.
- Mosey, N., et al., Interpretation of experiments on ZDDP anti-wear films through pressure-induced cross-linking. Tribology Letters, 2006. 24(2): p. 105-114.
- Bec, S., et al., Relationship between mechanical properties and structures of zinc dithiophosphate anti–wear films. Proceedings of the Royal Society of London. Series A: Mathematical, Physical and Engineering Sciences, 1999. 455(1992): p. 4181-4203.
- Ghanbarzadeh, A., et al., Zinc dialkyl dithiophosphate antiwear tribofilm and its effect on the topography evolution of surfaces: A numerical and experimental study. Wear, 2016. 362: p. 186-198, https://doi.org/10.1016/j.wear.2016.06.004.
- Dorgham, A., et al., On the transient decomposition and reaction kinetics of zinc dialkyldithiophosphate. ACS applied materials & interfaces, 2018. 10(51): p. 44803-44814.
- Mourhatch, R. and P.B. Aswath, Tribological behavior and nature of tribofilms generated from fluorinated ZDDP in comparison to ZDDP under extreme pressure conditions—Part II: Morphology and nanoscale properties of tribofilms. Tribology International, 2011. 44(3): p. 201-210.
- Parsaeian, P., et al., A new insight into the interfacial mechanisms of the tribofilm formed by zinc dialkyl dithiophosphate. Applied Surface Science, 2017. 403: p. 472-486, https://doi.org/10.1016/j.apsusc.2017.01.178.
- Vakis, A., et al., Modeling and simulation in tribology across scales: An overview. Tribology International, 2018. 125: p. 169-199, https://doi.org/10.1016/j.triboint.2018.02.005.


Be the first to comment